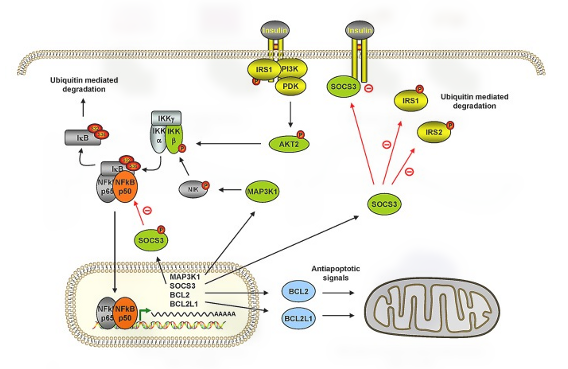
We are focusing in two major areas:
 We are also developing and applying computational tools to answer biomedical questions to better understand complex diseases by using all types of genetic and genomic data. Particularly, we have been working on generating efficient and accurate computational strategies to better understand the association between human genetic variability and complex diseases. For example we have developed and applied several strategies and computational methods to enhance the analysis of genomic information for genome-wide association studies (GWAS) in many ways. Among others, we have developed filtering schemes that minimize false positive associations from genotype imputation results and have applied them successfully to different complex diseases, such as Asthma and Type 2 Diabetes. We are also developing alternative strategies to enhance the association signals of large GWAS studies using gene and pathway information.
We are also developing and applying computational tools to answer biomedical questions to better understand complex diseases by using all types of genetic and genomic data. Particularly, we have been working on generating efficient and accurate computational strategies to better understand the association between human genetic variability and complex diseases. For example we have developed and applied several strategies and computational methods to enhance the analysis of genomic information for genome-wide association studies (GWAS) in many ways. Among others, we have developed filtering schemes that minimize false positive associations from genotype imputation results and have applied them successfully to different complex diseases, such as Asthma and Type 2 Diabetes. We are also developing alternative strategies to enhance the association signals of large GWAS studies using gene and pathway information.
A major current limitation of GWAS is the downstream interpretation of the functional role of the variants associated to the pathology. Our group, beyond identifying the genetic and molecular basis of disease, is also devoted to provide functionality to these variants, which are often located in apparently non-functional genomic regions. Using a combination of genotype imputation and Systems and Network biology approaches we aim to identify the potential causal variants, often not detected using a standard analytical approach, and to infer its potential functional impact. These approaches are already allowing us to identify candidate disease genes by using a combination of functional relationships together with gene-based tests.
 Cancer genomics covers a great part of our interest and research activity. In the context of different international consortia we are developing and applying analysis tools and approaches to shed light on the underlying genomic and functional basis of several types of tumors. In the race of understanding the biology of tumors and to find particular and personalized therapies, our contribution to the field is centered in the computational analysis of variation at different levels. In this context, we have recently developed SMuFin (for Somatic MUtation FINder), a reference-free method for the identification of genomic mutations that are responsible for the development and progression of tumors. By directly comparing the sequences of normal and tumor cells, SMuFin can identify (in a single run) nearly all types of somatic variants, from single nucleotide variations, to large structural changes implying complex rearrangements of chromosomes, e.g. those involved in chromotripsis and chomoplexy. With this, and other tools in hand, we aim to search for complex genomic landscapes that are associated to cancer and to identify their underlying mechanisms. We are also interested in uncovering, beyond the affected genes, to what extend the variation in gene regulatory regions can lead to oncogenesis. In this context, we will also use several tools developed in the group for the identification of promoters and enhancers in eukaryotic genomes (e.g. ReLA ). The combination of the identification of variants associated to tumor formation and the analysis of regulatory regions can allow us to find this missing link between gene regulation and cancer origin and progression.
Cancer genomics covers a great part of our interest and research activity. In the context of different international consortia we are developing and applying analysis tools and approaches to shed light on the underlying genomic and functional basis of several types of tumors. In the race of understanding the biology of tumors and to find particular and personalized therapies, our contribution to the field is centered in the computational analysis of variation at different levels. In this context, we have recently developed SMuFin (for Somatic MUtation FINder), a reference-free method for the identification of genomic mutations that are responsible for the development and progression of tumors. By directly comparing the sequences of normal and tumor cells, SMuFin can identify (in a single run) nearly all types of somatic variants, from single nucleotide variations, to large structural changes implying complex rearrangements of chromosomes, e.g. those involved in chromotripsis and chomoplexy. With this, and other tools in hand, we aim to search for complex genomic landscapes that are associated to cancer and to identify their underlying mechanisms. We are also interested in uncovering, beyond the affected genes, to what extend the variation in gene regulatory regions can lead to oncogenesis. In this context, we will also use several tools developed in the group for the identification of promoters and enhancers in eukaryotic genomes (e.g. ReLA ). The combination of the identification of variants associated to tumor formation and the analysis of regulatory regions can allow us to find this missing link between gene regulation and cancer origin and progression.
Beyond the application of these tools for answering specific biomedical questions, and given the potential of these tools, we also continue improving and developing further these methodologies in different directions. For example, we are interested in generating tools for the reconstruction of complex karyotypes, or to identify structural variants contributing to the variability at individual and at species level.